|
Variations in crude oil density are shown as function of temperatur, together with volume correction factors.ĭensities of Aqueous Solutions of Inorganic ChloridesĬhanges in density of aqueous solutions with changes in concentration at 20☌. 
Online API to Specific Gravity calculator.Ĭalculator and formulas for conversion between different units of concentration: Molarity, molality, mole fraction, weight percent of solute and grams of solute per liter of solution - descriptive terms for solubility. Molweight, melting and boiling point, density, pKa-values, as well as number of carbon and hydrogen atoms in molecules are given for 150 different alcohols and acids.ĪPI expresses the gravity or density of liquid petroleum products. 
Material properties of gases, fluids and solids - densities, specific heats, viscosities and more.Īlcohols and Carboxylic Acids - Physical Data See also density of aqueous solutions of organic acids, inorganic chlorides, inorganic sodium salts, inorganic potassium salts and some other inorganic substancesĭensity of aqueous solutions at 20☌, given as g/cm 3:įor full table with Glycerol, b-D-Lactose, a-Maltose, D-Mannitol, Methanol, 1-propanol, 2-propanol, Sucrose and Urea - rotate the screen! Aqueous Solutions of Organic Substances as Sugars and Alcohols - Densities Mass%ĭensity of aqueous solutions at 20☌, given as wt%Ĭonversion of the concentration from mass% to mol/kg (moles of solute/kg of water = molality):įor full table with Glycerol, b-D-Lactose, a-Maltose, D-Mannitol, Methanol, 1-propanol, 2-propanol, Sucrose and Urea - rotate the screen! Aqueous Solutions of Organic Substances as Sugars and Alcohols - Densities - Conversion of the concentration from mass% to mol/kg Mass%Ĭonversion of the concentration from mass% to mol/liter (moles of solute/liter of solution = molarity):įor full table with Glycerol, b-D-Lactose, a-Maltose, D-Mannitol, Methanol, 1-propanol, 2-propanol, Sucrose and Urea - rotate the screen! Aqueous Solutions of Organic Substances as Sugars and Alcohols - Densities - Conversion of the concentration from mass% to mol/liter Mass%ĭensities of solids, liquids and gases. Mol/liter: Molarity = moles of solute/liter of solution Mol/kg: Molality = moles of solute/kg of water Wt%: Mass of solute/total mass of solution*100% 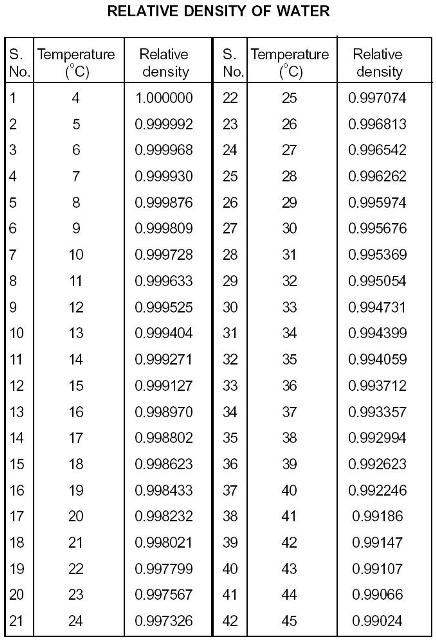
Be aware of the concentration units in the figures:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
